Summary
The FDA has proposed phasing out oral phenylephrine, a common decongestant in cold medicines like Sudafed PE and DayQuil, after studies showed it is no more effective than a placebo.
The drug, ineffective when swallowed due to breakdown in the stomach, remains usable in nasal sprays.
Alternatives include pseudoephedrine, nasal sprays, and steroid treatments like Flonase.
The regulatory process to remove phenylephrine could take over a year, but experts argue removing ineffective options will help consumers choose better remedies for congestion. Drugmakers are expected to challenge the proposal.


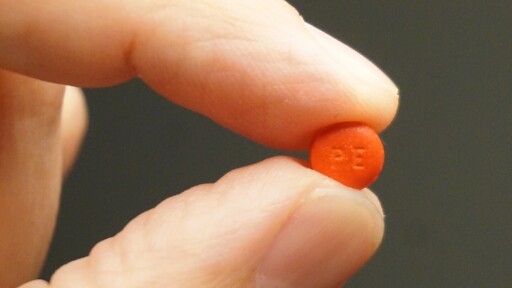
The stuff that they restricted.